Research Funding:
Overall, I have received ~$2.0 million in funding across topics of ocean sciences, experimental evolution, pedagogy, DEI, and student success. My current research focuses on the evolutionary responses of marine zooplankton to climate change stressors. We use an experimental evolutionary approach to tease apart ecologically plastic interactions and evolutionary changes and constraints. I am co-author on several high-impact publications in journals like Nature Climate Change, Nature Communications, Proceedings Royal Society B, and Proceedings of the National Academy of Sciences.
Michael Finiguerra (PI), Rachael Gabriel (Co-PI; NEAG School Education). 2016-2018 Integrating research and practice in promoting coastal literacy in high school classrooms: A collaborative approach among teachers, education researchers, and coastal scientists Connecticut Sea Grant $150,000; (this is the first pedagogy proposal ever funded by CT Sea Grant).
Hans Dam (PI, Marine Sciences), Michael Finiguerra (Co-PI), Hannes Baumann (Co-PI; Marine Sciences). 2016-2018 Predicting the performance of the copepod Acartia tonsa under future conditions of temperature and CO2. Connecticut Sea Grant $150,000
Hans Dam (PI, Marine Sciences), Michael Finiguerra (Co-PI), Hannes Baumann (Co-PI; Marine Sciences).2016-2019 Collaborative research: Transgenerational phenotypic and genomic responses of marine copepods to the interactive effects of temperature and CO2 National Science Foundation- Biological Oceanography ~$522,302
Hans Dam, Michael Finiguerra “The Effect of Timing and Duration of Climatic Heat Waves in Long Island Sound to a Zooplankton Population.”*CT Sea Grant: 2/14- 1/16 $ 128,732
Hans Dam, Michael Finiguerra “Collaborative Research: Costs and Advantages of a Novel Sodium Channel Mutation in Copepods”* National Science Foundation- Integrative Organismal Systems 3/10-3/13 $ 571,876
*Co-author of proposal as a graduate student; funded PhD research
I am a broadly trained integrative biologist. I have expertise in many facets of microbiology, molecular biology, and evolutionary ecology (e.g., viral isolation and infectivity, gene expression, life-history experiments). My research specializes in the molecular ecology of microplankton; connecting measurements of fi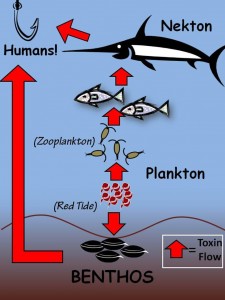 tness (e.g., reproductive rates, growth efficiencies) with gene expression. I aim to understand the molecular mechanisms behind adaption to environmental stressors (toxins, temperature, etc.); whether these mechanisms are sensitive to natural selection or plastic in response. I use an easily cultured toxic unicellular algae-coastal copepod as a model system and employ a wide range of molecular and ecological methods to address these questions. My work is hypothesis-driven and employs a combination of field and lab studies. I am an extremely motivated, enthusiastic researcher who accomplishes goals and secures funding. Further, I have helped form collaborations and am an excellent team member.
tness (e.g., reproductive rates, growth efficiencies) with gene expression. I aim to understand the molecular mechanisms behind adaption to environmental stressors (toxins, temperature, etc.); whether these mechanisms are sensitive to natural selection or plastic in response. I use an easily cultured toxic unicellular algae-coastal copepod as a model system and employ a wide range of molecular and ecological methods to address these questions. My work is hypothesis-driven and employs a combination of field and lab studies. I am an extremely motivated, enthusiastic researcher who accomplishes goals and secures funding. Further, I have helped form collaborations and am an excellent team member.
My research interests are:
1. Experimental Evolution in Copepods:
The effects of climate change on marine life are complex and often unknown. Many studies expose current phenotypes to future conditions, but ignore evolutionary change. We are interested in seeing how the effects of climate change, warming, acidification, and both, affect evolutionary trajectories in marine copepods. Copepods are a foundational organisms that are the link between primary production and higher trophic levels. Understanding if they can adapt, and if there is a cost to this adaptation, is critical to prediction how marine food webs will respond to climate change. No copepods, no fish or whales!
2. Evolutionary Ecology of Toxic Algal Blooms:
Toxic algal blooms are increasing in occurrence and intensity worldwide, and pose serious human health and economic concerns. Yet, the mechanisms that control their growth are poorly understood. My main research focus investigates the role of grazer control of toxic phytoplankton. Locally, the toxic dinoflagellate Alexandrium sp. has re-occurred annually in Northport Harbor, western Long Island Sound, since 2006. My dissertation research tested the hypothesis that a novel sodium channel isoform confers tolerance to Alexandrium neurotoxins in the copepod Acartia hudsonica (a dominant zooplankton grazer). I measured the advantages, costs, trade-offs, and population dynamics associated with the mutant isoform under toxic (blooms of Alexandrium) and non-toxic conditions. I pioneered the technique of measuring fitness and gene expression on the same individual copepod. I was a co-author on the NSF-IOS grant that funded my dissertation research. I also participated in a two-year study to identify the role of grazing on toxic and non-toxic cells during the progression of the toxic Alexandrium blooms in Northport Harbor, done in collaboration with Stony Brook University. In this project, we were able to calculate the impact of protistan and metazoan plankton grazing on phytoplankton and, specifically, Alexandrium populations. This project used classical field ecology methods with cutting edge molecular techniques. Lastly, because Alexandrium toxins kill grazers through physiological incapacitation leading to starvation, and because males appear more susceptible to Alexandrium, we tested the hypothesis that male copepods are more susceptible to starvation than females. In addition to pursuing my own interests in this area, I foresee a continued collaboration with Dr. Dam’s group.
This video illustrates how toxic Alexandrium spp. affects the copepod Acartia hudsonica. This is a time series of the same copepod before and after feeding on toxic cells for 24 hours.
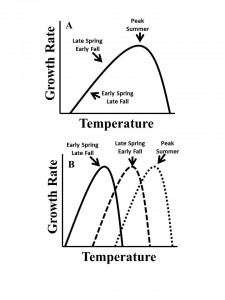
3. The Evolutionary and Ecological Consequences of Timing and Duration of Heat-Waves on Coastal Zooplankton:
As a result of climate change, the frequency and duration of extreme climate events, such as droughts, floods, and heat waves, are projected to increase worldwide. Dr. Dam and I co-authored the CTSG proposal that will investigate the eco-evolutionary effects of heat-waves on the copepod community in Long Island Sound. We will first determine if copepods have fixed or seasonally-variable thermal performance curves. Then, the evolutionary and ecological consequences (e.g., bottleneck or mis-match with fish larvae predators, respectively) of the timing and duration of heat-waves on copepods will be examined.
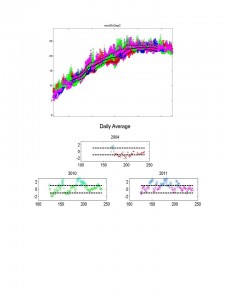
Top:Temperature for the period April- Sept. for 2004-2011 in the main basin of LIS. Colors represent different years for data collected every 15 minutes. The black lines represent the mean climatological daily temperature and 1 standard deviation above the means. Not all years are visible. Courtesy of Dr. J.O’Donnell. Data from LISICOS EXRK buoy data. www.lisicos.uconn.edu
Bottom: Daily average temperatures anomalies (Y Axis) versus day of the year for selected years in LIS. Dotted lines = + 1 SD around mean anomaly of zero. Positive anomalies represent warmer-than- average-days. Days with positive anomalies > 1 SD from the mean are circled.
3. Marine Viral Ecology:
For my Master’s thesis I examined the effects of inorganic and organic colloids and molecules, exposures to non-receptive bacterial cells and extracellular enzymes on virion decay. I also participated in numerous research cruises that determined the spatial distribution of viral, bacterial, and sewage contaminant abundances in Long Island Sound. I an excited to continue my marine microbial research.
4. Methyl-Mercury Dynamics in Zooplankton:
Methyl-mercury is a global toxic pollutant that causes severe human health problems. Methyl -mercury propagates from phytoplankton up the food web. I have been collaborating with Dr. W. Fitzgerald (UConn) and Dr. C. Hammerschmidt (Wright State University) on generating the first open ocean measurements of methyl-mercury in zooplankton. On three oceanographic cruises to the continental shelf of the northwest Atlantic, I collected size-fractionated zooplankton and phytoplankton samples to be analyzed by Dr. Hammerschmidt for methyl -mercury. I modified plankton sampling equipment and ecological experiments to adhere closely to trace metal clean protocols. These samples will aid in the understanding of the spatial distribution, bioaccumulation, and biomagnification of methyl-mercury in plankton.